|
Links |
Are there allelic variants in human population that are protective against HIV?
|
|
|
What is MHC and what does it do? What is the molecular basis of chimpanzee resistance to SIVcpz? Are there allelic variants in human population that are protective against HIV? How are humans adapting to selection pressures produced by HIV? What will the future bring?
|
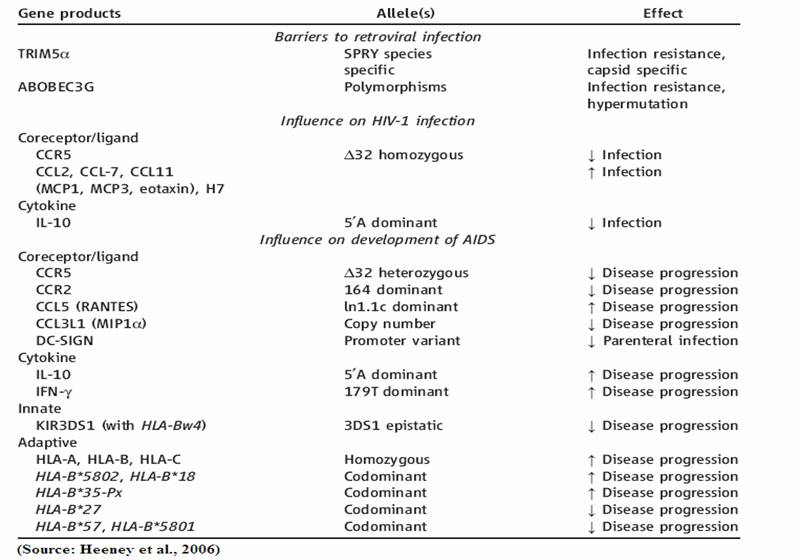
Within the human population a number of polymorphisms exist that block or slow the progression of the HIV-1 virus. At present there are 10 genes and 14 alleles known to have either a beneficial or detrimental effect on HIV-1 infection and/or disease progression (3; 7). The table below provides a current list of these genes and allelic variants that have a bearing on HIV infection and disease. For example, the CCR5-∆332 allele restricts the entry of HIV-1 into white blood cells. It should be noted that although positive natural selection may be the explanation provided for the relatively high frequency of the CCR5-∆332 allele in some European populations, the selection pressure was not caused by the HIV virus. This is said because the HIV epidemic is a fairly new phenomenon thus positive selection pressure was probably a on another allele that exists in linkage disequilibrium with the CCR5- ∆332 variant (12).
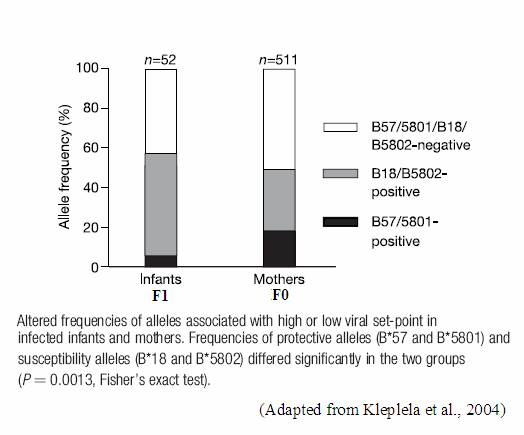
The different classes of HLA do not contribute equally to the immune recognition of, and response to HIV-1 (4). The contribution of HLA-B alleles is far greater than that of both HLA-A and HLA-C alleles, thus it is important to consider the strong differential advantage or disadvantage that each particular classes of HLA confer during an HIV infection. Additionally, it is necessary to consider the selective benefits or deficits associated with each allele within each of these HLA classes. In particular, specific HLA-B alleles have been identified as having a profound influence on HIV-1 viral loads (3; 4; 5). Depending on the HLA-B alleles present, a higher or lower viral load may be observed. Furthermore, in an individual infected with HIV-1 homozygozity at the HLA-B loci is proven to be far more deleterious than homozygozity at the HLA-A locus, as the relative rate of progression to AIDS is 2-3 times greater in HLA-B homozygotes (4). Specifically, HLA-B*57 and HLA-B*801 are associated with slower disease progression in infected individuals. Conversely, HLA-B*18 and HLA-B*5802 are coupled with the rapid progression of HIV to AIDS (3; 4). A possible mechanism for the dominance of HLA-B is the greater diversity of peptides that can be bound by HLA-B alleles (4). Perhaps the larger degree of functional diversity seen in HLA-B, relative to HLA-A, slows the process of adaptation of the HIV-1 virus. Due to the greater amount of functional diversity associated with HLA-B, HLA-B alleles will undoubtedly continue to be the focal point of the selection pressure imposed by the HIV-1 epidemic (4). However, it is important to note that the specific alleles that are selected for may change as the HIV-1 epidemic progresses and the virus diversifies and adapts (4). This phenomenon is illustrated in the figure to the below. Although the overall frequency of positively contributing alleles that are present in the F1 generation increases, the frequency of the positive allele that predominated in the F0 generation decreased. Nonetheless, this decline was compensated by an increase in frequency of an alternate positive allele. Additionally, the frequency of negative (non-protective) alleles decreases in the population over the course of one generation, thus further illustrating the strength of the selection pressure exerted by HIV-1 on the HLA-B locus (4).
|